  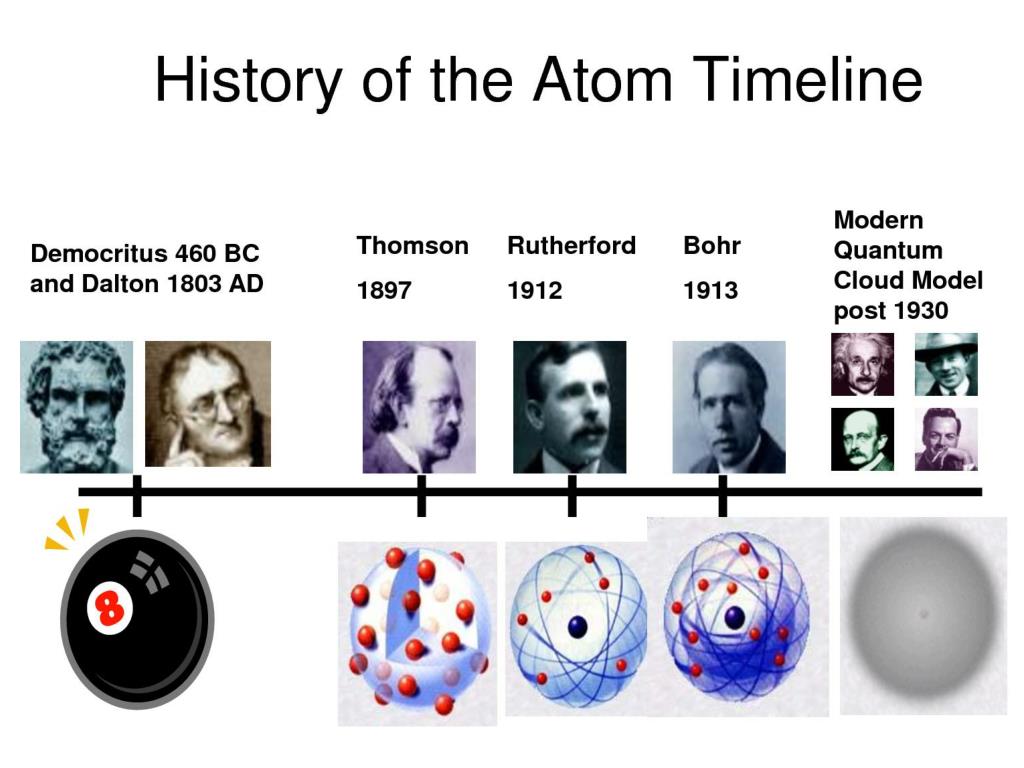
Their speculation about a hard, indivisible fundamental particle of nature was replaced slowly by a scientific theory supported by experiment and. Through different experiments with gases Dalton expanded on this to theorise that atoms vary in size and mass and that compounds had to be made of whole number ratios of atoms. The concept of the atom that Western scientists accepted in broad outline from the 1600s until about 1900 originated with Greek philosophers in the 5th century bce. Scientists have taken the hypothesis of Greek scholars and have built on it with their different discoveries and theories regarding the atom, which derives from the Greek word 'atomos,' meaning indivisible. 
This built on the work of Lavoisier and Dalton furthered this proposing that each chemical element is made of atoms of a unique type and they cannot be altered or destroyed but can be combined. Best Known For: Chemist John Dalton is credited with pioneering modern atomic theory. Dalton’s experiments with gases led to some of the earliest measurements of atomic masses and a concept of atomic structure and reactivity. Atomic theory has evolved since ancient times. He found that 100g of tin will combine with 13.5g or 27g of oxygen and that this could be represented by a 2:1 ratio, for every 2 atoms of oxygen there was one atom of tin. He looked at tin oxide and the combination of masses of oxygen with tin. He also researched colour blindness, which he had as a result, colour blindness is known as Daltonism in several languages. 1 He introduced the atomic theory into chemistry. Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford. John Dalton FRS ( / dltn / 5 or 6 September 1766 27 July 1844) was an English chemist, physicist and meteorologist. Outline milestones in the development of modern atomic theory. Dalton used the work of Lavoisier and Joseph Proust to examine the ratios of elements that combine to form compounds and look at their ratio of masses. Use postulates of Dalton’s atomic theory to explain the laws of definite and multiple proportions. The work of Lavoisier and the atomists was furthered in the 18th Century by the British scientist John Dalton. It also proved the earlier work of Robert Boyle who hypothesised in 1661 that elements cannot be broken down into simpler substances. This was a crucial breakthrough in the work of atomists in confirming what matter was made of as it was proved that atoms are not created or destroyed when a reaction happens. This led to the theory of the law of conservation of mass. Rutherford model, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. They formulated the key concepts of the law of conservation of mass and the existence of atoms as the building blocks of all matter using their knowledge of chemical reactions.Ī later breakthrough in the discovery of the atomic model came through the work of French chemist Antoine Lavoisier who through a series of experiments found that the total mass of products and reactants in a chemical reactions is always the same. The modern Atomic Model was first developed by two key scientists Lavoisier and Dalton with the help of others. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
